Development of the Commercial Manufacturing Process for Ipatasertib
Ipatasertib is a potent small molecule Akt kinase inhibitor currently tested in Phase III clinical trials for the treatment of metastatic castration-resistant prostate cancer and triple negative metastatic breast cancer. In this presentation an overview of the development activities towards the commercial manufacturing process is given. The convergent synthesis comprises ten steps with eight isolated intermediates and utilizes a wide range of chemical techniques and technologies to build this complex drug. All three stereocenters are introduced using enzyme as well as metal catalysis.
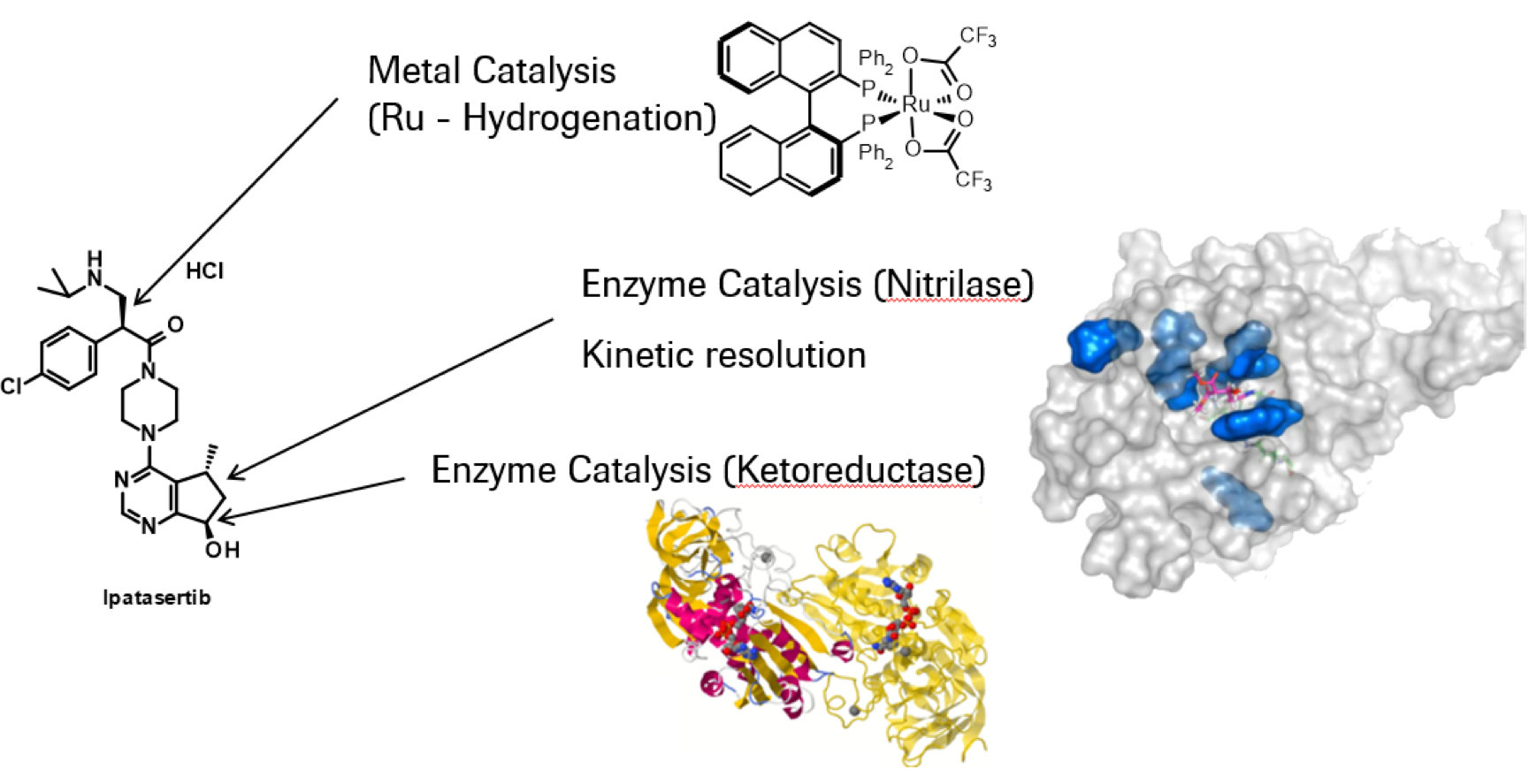
[1] Remarchuk, S. Babu, J. Stults, A. Zanotti-Gerosa, S. Roseblade, S. Yang, P. Huang, C. Sha, and Y. Wang, Org. Process Res. Dev. 2014, 18, 1641−1651.
[2] Remarchuk, F. St-Jean, D. Carrera, S. Savage, H. Yajima, B. Wong, S. Babu, A. Deese, J. Stults, M. W. Dong, D. Askin, J. W. Lane, and K. L. Spencer, Org. Process Res. Dev. 2014, 18, 1652−1666.